Abstract
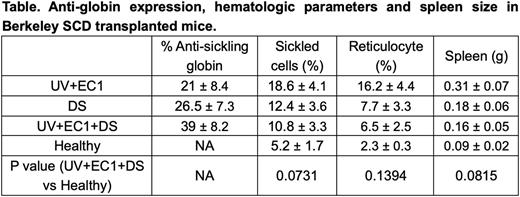
Sickle cell disease (SCD) is one of the most common genetic blood diseases. Multiple promising gene therapy approaches are being tested, including inducing fetal hemoglobin (HbF) production through reducing expression of the HbF repressor BCL11A and introducing anti-sickling beta-globin variants. While current gene therapy strategies focus on single mechanisms to mitigate the SCD phenotype, we developed a therapeutic approache combining an optimized lentivirus (LVV) expressing an anti-sickling betaAS3-globin gene (UV+EC1) and two shRNAs embedded in microRNAs (shmiRs) simultaneously targeting BCL11A and ZNF410 (UV+EC1+DS). Our goal was to maximize overall anti-sickling globin content through simultaneous expression of recombinant anti-sickling globin (AS3) and induction of endogenous anti-sickling HbF while simultaneously reducing sickle globin. We inserted a double shmiR targeting BCL11A and ZNF410 (DS) into the UV+EC1 vector, which is significantly reduced in size and supports increased titers and gene transfer to CD34+ HSPC, to engineer the novel vector, UV+EC1+DS. Healthy human HSC-derived erythroid cells transduced with the UV+EC1+DS LVV showed up to 27% AS3 globin and 20% HbF, resulting in a cumulative 47% anti-sickling globin. Erythrocytes differentiated from HSCs derived from patients with SCD and transduced with the UV+EC1+DS expressed therapeutic levels of anti-sickling globin (AS3+HbF) of 44%, significantly higher than either UV+EC1 or DS transduced cells, and reduced in vitro sickling from 60% to 15%, significantly lower than UV+EC1 or DS alone (P<0.001). Berkeley SCD mice engrafted with LVV-transduced cells demonstrated an average level of anti-sickling globin in erythroid cells from BM of 21%, 27% and 39% for UV+EC1, DS and UV+EC1+DS, respectively (Table). This increase in anti-sickling globin was associated with a statistically significant reduction in peripheral blood hemolysis markers, and the UV+EC1+DS group showed red cell parameters and spleen sizes that were not significantly different from mice which received healthy donor cells. These data highlight the potential of a novel combination vector using two distinct mechanisms to ameliorate SCD. The new vector can improve the efficacy of current gene approaches by combining different therapeutic strategies.
Disclosures
Kohn:Cimeio Therapeutics: Consultancy; Innoskel: Consultancy; MyoGene Bio: Consultancy, Other: Membership on Scientific Advisory Board; Pluto Immunotherapeutics: Consultancy, Other: Membership on Scientific Advisory Board; Allogene Therapeutics: Consultancy, Other: Member of Scientific Advisory Board; ImmunoVec: Consultancy; TransformaTx: Consultancy. Williams:Insertion Site Advisory Board, Biomarin: Consultancy; Insertion Site Analysis Advisory Board, Bluebird Bio: Consultancy; Scientific Advisory Board, Beam Therapeutics: Consultancy; Bluebird: Consultancy, Other: Provides Vector; Chief Scientific Chair, Emerging Therapy Solutions: Consultancy; Scientific Advisory Board, Skyline Therapeutics: Consultancy; Novartis: Consultancy, Other: Steering Committee (fees donated to NAPAAC); Orchard Therapeutics: Other: Provides vector; Novartis: Other: Provision of study materials, medical writing.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal